Cytokine storm assay services for immunogenicity risk assessment
Cytokine storm assays are widely implemented to assess immunogenicity risk at an early stage. Uncontrolled cytokine release activation can lead to severe inflammatory responses and has been associated with serious adverse events during clinical development.
At BioMedha, we perform cytokine storm assays using human donor PBMCs to assess immunogenicity risk in vitro at an early stage.
Cytokine storm assays are widely used during drug discovery to evaluate the potential risks of therapeutic candidates to trigger excessive immune activation. Uncontrolled cytokine release can lead to severe inflammatory responses and has been associated with serious adverse events during clinical development.
At BioMedha, we perform cytokine storm assays using human donor PBMCs to investigate immune activation and assess immunogenicity risk in vitro. These in vitro assays are suitable as a predictive model for immunogenicity risk assessment (cytokine storm). Assay utilizes Luminex multiplexing magnetic bead based assay detection help drug developers evaluate whether therapeutic molecules may induce cytokine release at an early stage.
Why cytokine storm assays are important in drug development
Biologics, therapeutic proteins, and immune-modulating drugs can trigger excessive immune responses known as cytokine storm. Early identification of this risk is critical to ensure patient safety and improve drug development success.
In vitro cytokine storm assays allow researchers to:
Assess immunogenicity risk posed by therapeutic candidates
Support early safety decision-making in drug discovery
These assays are particularly relevant for biologics, antibodies, drug conjugates, and immune-modulating small molecules, where immune activation must be carefully controlled.
Cytokine storm assay platform at BioMedha
BioMedha performs in vitro cytokine storm assays using peripheral blood mononuclear cells (PBMCs) isolated from healthy donor whole blood.
The assay measures cytokine secretion following exposure to test compounds, providing insight into immune activation and inflammatory signalling.
Key features of the assay include:
PBMCs isolated from healthy donor whole blood within 2 hours of blood collection
Multi-donor testing to evaluate biological variability
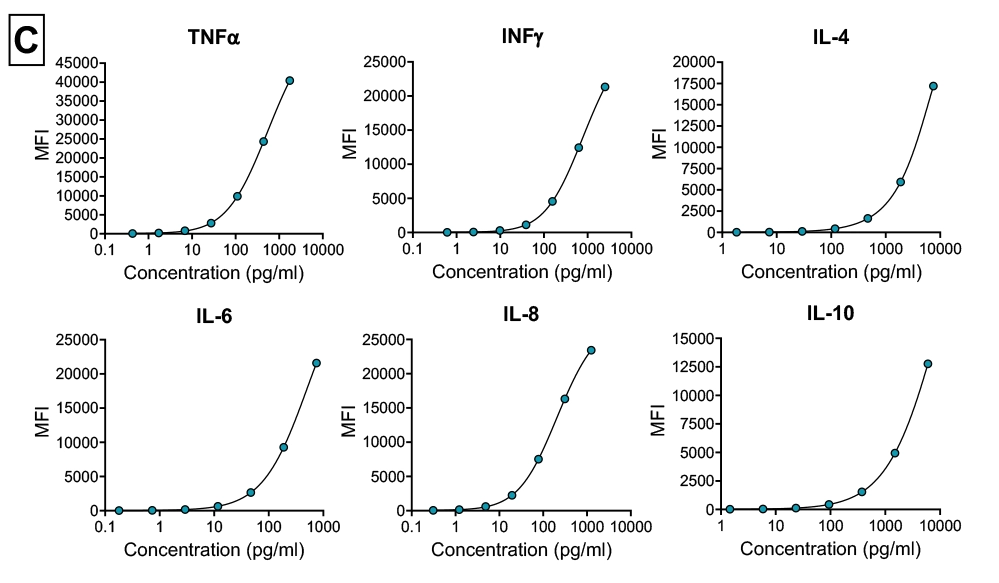
Detection using Luminex multiplex magnetic bead assays
Simultaneous measurement of multiple cytokines
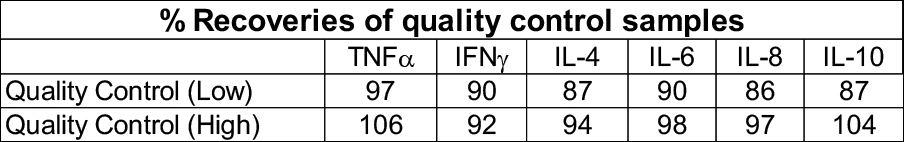
Quantitative analysis of cytokine induction compared with vehicle controls
These assays provide a robust platform for assessing immunogenicity risk and immune safety profiling during early development.
Example cytokine storm assay case study
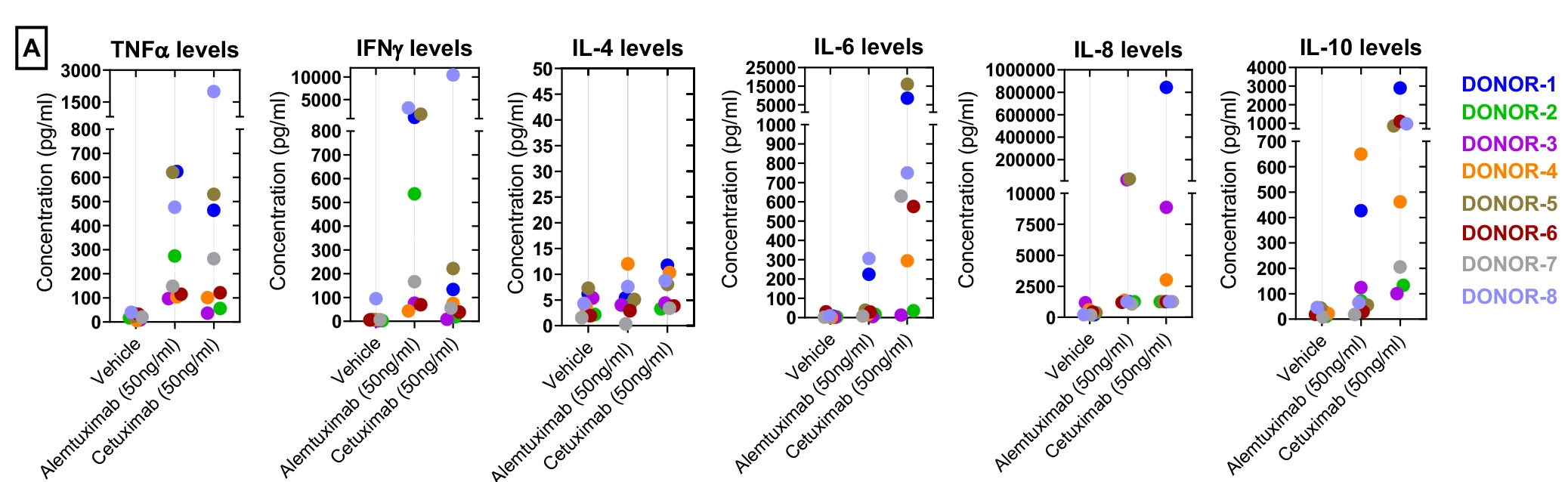
The following example summarises a cytokine storm assay performed using PBMCs from eight healthy donors.
Cytokine secretion concentration analysis
Figure-A shows the concentration of cytokine analytes (pg/mL) measured in PBMC supernatant following compound exposure.
Data were plotted with cytokine concentration on the y-axis and test conditions on the x-axis. Each donor is represented by a distinct colour code, and each data point represents the average of duplicate wells.
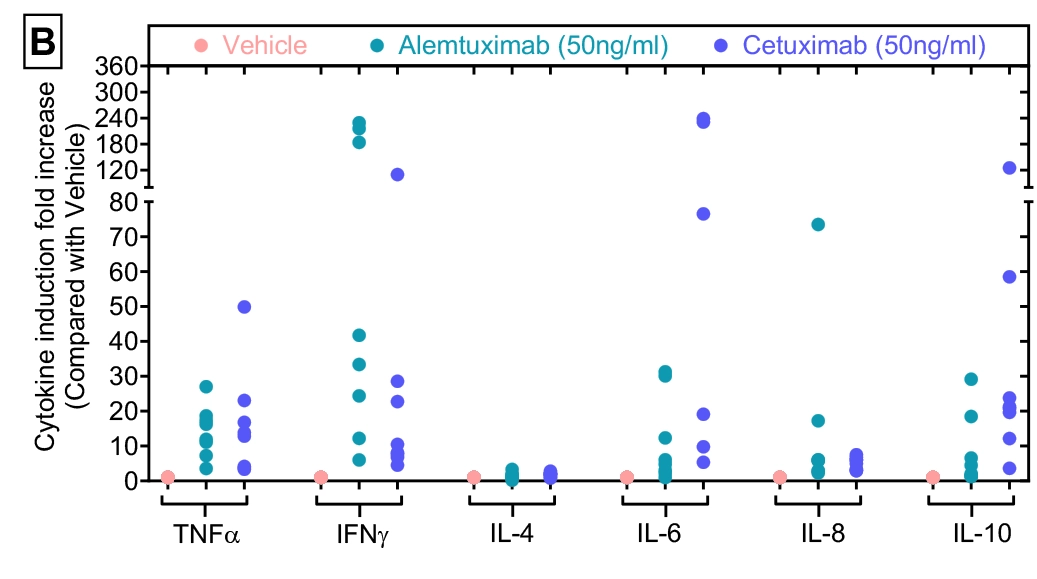
Cytokine induction analysis
Figure-B illustrates cytokine induction fold increase relative to vehicle control across eight donors.
The cytokine concentration data shown in Figure A were re-analysed to calculate fold induction in response to test agents, enabling comparison of immune activation across different experimental conditions.
Example cytokine storm assay case study
The response exhibited by Alemtuzumab and Cetuximab in this assay agrees with clinical immunogenicity findings in the literature. In vitro Cytokine storm assays developed at BioMedha are robust tools for implementing and assessing immunogenicity risk at an early stage. Understanding and managing immunogenicity at the earliest possible stage is important to improve safety and efficacy of therapeutic molecules in drug development.
Related immunology and toxicology assays
- Whole blood assays
- T Cell assays
- PBMC assays
- Neutrophil assays
- Immune cell mediated tumour killing
- Biomarker analysis
- Cytokine assays
- Flow cytometry
Discuss your immunogenicity assessment study
BioMedha provides specialist immunology CRO services supporting early-stage drug discovery and immune safety research.
If you are evaluating compounds for immunogenicity risk, our scientists can help design and impliment the study aligned with your research objectives.