Mitochondrial toxicity assay services for safety assessment at an early stage
Mitochondrial toxicity is a major contributor to drug-induced adverse effects and is an important factor in the failure of drug candidates during development. Damage to mitochondrial function can disrupt cellular energy production, leading to cell dysfunction or death.
At BioMedha, we provide mitochondrial toxicity assay services designed to identify early signs of mitochondrial dysfunction during preclinical drug discovery. Our assays enable researchers to evaluate how compounds affect mitochondrial activity, cellular metabolism and overall cell viability.
These studies help drug discovery teams detect drug-induced mitochondrial toxicity early, reducing the risk of safety issues later in development.
Why mitochondrial toxicity testing matters in drug discovery
Mitochondria are responsible for producing the majority of cellular energy in the form of ATP and play a central role in metabolism, signalling and apoptosis. Disruption of mitochondrial function can therefore lead to significant cellular damage and has been linked to many cases of drug-induced toxicity.
Screening compounds for mitochondrial toxicity assessment during early development allows researchers to:
Identify mitochondrial toxicity liabilities in compounds
Understand mechanisms of drug-induced toxicity
Improve compound selection during lead optimisation
Reduce the risk of late-stage safety failures
For many drug discovery programmes, mitochondrial toxicity screening is now a key component of early safety assessment.
Mitochondrial toxicity assays at BioMedha
BioMedha provides in vitro mitochondrial toxicity assays to support early safety evaluation of drug candidates.
Our assays are designed to detect compound-induced mitochondrial dysfunction by assessing multiple cellular and metabolic parameters.
Typical measurements include:
- Cellular ATP production in the presence of glucose/ galactose media
- Mitochondrial membrane potential (MMP assay)
- Reactive oxygen species (ROS) generation
- Mitochondrial respiration and metabolic activity
- Cell viability and cytotoxicity
By analysing these parameters, we can determine whether compounds interfere with mitochondrial energy production, or disrupt mitochondrial membrane potential or induce mitochondrial stress.
Figure-A: Measuring ATP generation in the presence of glucose (glucose media) and absence of glucose (galactose media). Cells treated with tool compounds and ATP levels measured using ATPlite luminescence readout. Percent (%) ATP levels calculated by normalisation to vehicle response. Culturing cells in glucose media favour glycolysis over oxidative phosphorylation for producing ATP. Cells cultured in absence of glucose, forces cells to generate ATP by oxidative phosphorylation there by we can identify compounds which cause mitochondrial impairment.
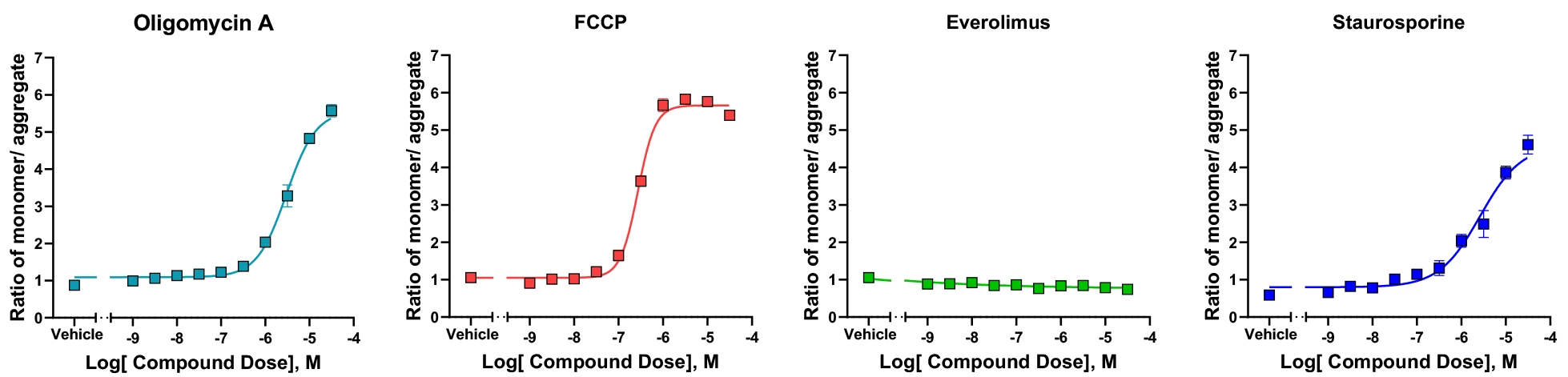
Membrane Potential Assay
Figure-B: Mitochondrial Membrane Potential (MMP) assay utilises JC-10 dye for measuring mitochondrial depolarization in cells. JC-10 is a cationic dye accumulates in energised mitochondria. Cells treated with tool compounds. At high membrane potentials, the dye aggregates and at low membrane potentials the dye exists as a monomer. Ratio of monomer/aggregate JC-10 fluorescence intensity was measured as an indicator of mitochondrial impairment.
Identifying potential mitochondrial toxins as early as possible in drug discovery is crucial. Here we demonstrate sensitive assay design approach as an indicator to assess drug-induced mitochondrial toxicity. Compound IC50 was derived from Dose Response curves in ATP synthesis assay and Mitochondrial Membrane Potential (MMP) assay.
Why mitochondrial toxicity testing matters in drug discovery
Mitochondria are responsible for producing the majority of cellular energy in the form of ATP and play a central role in metabolism, signalling and apoptosis. Disruption of mitochondrial function can therefore lead to significant cellular damage and has been linked to many cases of drug-induced toxicity.
Screening compounds for mitochondrial toxicity during early development allows researchers to:
Identify mitochondrial toxicity liabilities in compounds
Understand mechanisms of drug-induced toxicity
Improve compound selection during lead optimisation
Reduce the risk of late-stage safety failures
For many drug discovery programmes, mitochondrial toxicity screening is now a key component of early safety assessment.
Integrated discovery toxicology capabilities
Mitochondrial toxicity assessment at BioMedha is supported by our broader Discovery Toxicology services, allowing researchers to investigate compound safety using complementary assays.
Related toxicology assays may include:
Cytotoxicity assays
Oxidative stress assays
hERG binding assay
DNA damage assay
Integrating mitochondrial toxicity assays with other toxicology approaches enables a more comprehensive evaluation of compound safety.
Related assay services
- Immune cell assays
- Target expression studies
- Discovery toxicology assays
- Flow cytometry
- Immunology studies
- Immuno-oncology assays
Discuss your mitochondrial toxicity study
BioMedha provides specialist preclinical CRO services supporting early drug discovery and toxicology research.
If you are evaluating compound safety or investigating mitochondrial dysfunction in your research programme, our scientists can help design a study aligned with your scientific objectives.